|
In the tenth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the ninth B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the eighth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There is one shorter (1.76 Å) and one longer (1.77 Å) B–B bond length. In the seventh B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. However, its ideal electron pair geometry is tetrahedral. There is one shorter (1.75 Å) and one longer (1.79 Å) B–B bond length. The molecular geometry or the shape of hydrogen sulfide (H 2 S) is bent, angular or V-shaped. 
In the sixth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fifth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the fourth B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the third B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. In the second B site, B is bonded in a 9-coordinate geometry to six Mg and three B atoms. In the first B site, B is bonded in a 9-coordinate geometry to one Li, five Mg, and three B atoms. There are a spread of Mg–B bond distances ranging from 2.48–2.52 Å. 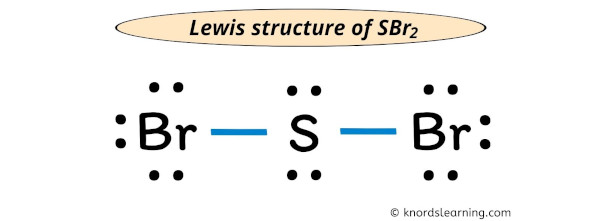
Determine the molecular geometry for each molecule Determine the idealized bond angles for each molecule. How many electron groups are on the central atom, Determine the electron geometry for each molecule. In the fifth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. Study with Quizlet and memorize flashcards containing terms like Draw the Lewis structure for CO., A molecule with the formula AB3 has a trigonal planar geometry. There are a spread of Mg–B bond distances ranging from 2.47–2.51 Å. In the fourth Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.48–2.51 Å. In the third Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share an edgeedge with one LiB12 cuboctahedra, edges with eleven MgB12 cuboctahedra, a faceface with one LiB12 cuboctahedra, and faces with seven MgB12 cuboctahedra. There are a spread of Mg–B bond distances ranging from 2.49–2.51 Å. The SBr2 molecule has a tetrahedral or V-shaped bent molecular geometry because. In the second Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with three equivalent LiB12 cuboctahedra, edges with nine MgB12 cuboctahedra, and faces with eight MgB12 cuboctahedra. There are a spread of more » Mg–B bond distances ranging from 2.48–2.50 Å. In the first Mg site, Mg is bonded to twelve B atoms to form MgB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra, faces with two equivalent LiB12 cuboctahedra, and faces with six MgB12 cuboctahedra. 
There are a spread of Li–B bond distances ranging from 2.48–2.50 Å. Li is bonded to twelve B atoms to form LiB12 cuboctahedra that share edges with twelve MgB12 cuboctahedra and faces with eight MgB12 cuboctahedra. The molecular geometry will thus be linear, the basic #"AX"_2# model.LiMg9B20 is hexagonal omega structure-derived structured and crystallizes in the triclinic P-1 space group. It will use one s and one p orbitals to form the hybrids, and the remaining p-orbitals to form pi bonds with the two sulfur atoms. The carbon atom will thus be #"sp"# hybridized. This means that its steric number will be equal to #2#. In this case, the carbon atom is surrounded by two regions of electron density, one for each double bond it forms with the sulfur atoms. Now, molecular geometry is determined by the hybridization of the central atom. 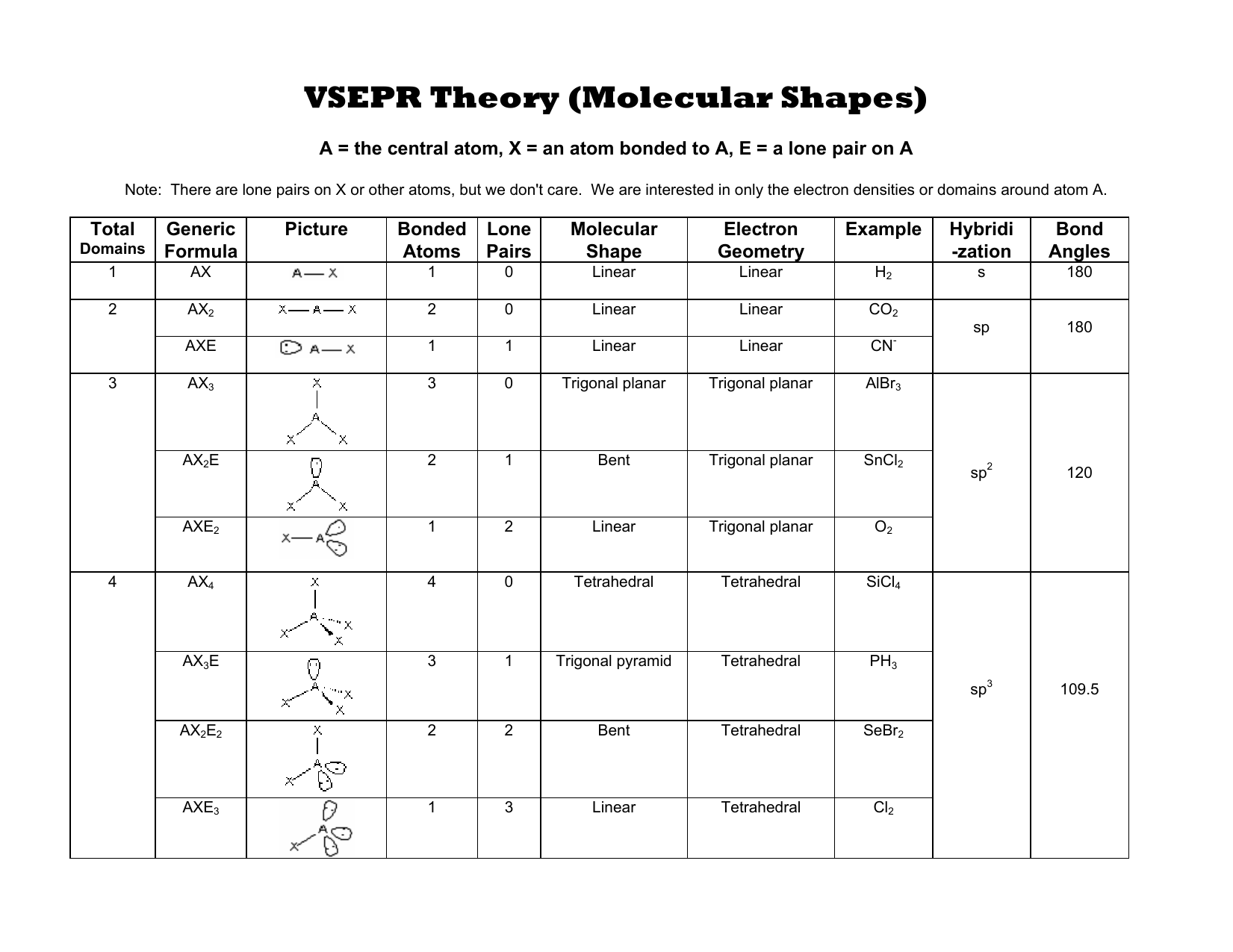
The remaining #8# valence electrons will be placed as lone pairs, two on each sulfur atom. These bonds will account for #8# of the #16# valence electrons of the molecule. The central carbon atom will form double bonds with the two sulfur atoms. The best place to start when trying to figure out a molecule's geometry is its Lewis structure.Ĭarbon disulfide, #"CS"_2#, will have a total of #16# valence electrons, #4# from the carbon atom and #6# from each of the two sulfur atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
